Water pumps with mechanical drive
Cooling Liquid
The cooling liquid is the transport medium that is used to transport the waste heat of the engine to the radiator or the heating cooler. Special cooling liquid compositions make a significant contribution to the proper function of the cooling system. In liquid-cooled vehicle engines – with a few exceptions, such as oil cooling – the cooling liquid consists of a mixture of water and coolant agent.
In terms of the function and the task, the correct cooling liquid is as important as the engine oil. Incorrect specifications, an unsuitable mix ratio, irregular replacement of the cooling liquid and/or ageing of the cooling liquid lead to corrosion and premature failure of the water pump and other engine parts. The additives in the coolant agent function as ageing stabilisers, corrosion protection, anti-foam agent, detergents and coating material. All additives ensure the proper function and condition of the cooling liquid in accordance with the regulations until the next change.
Some of the most important functions and facts regarding coolant agent are stated below.
In terms of the function and the task, the correct cooling liquid is as important as the engine oil. Incorrect specifications, an unsuitable mix ratio, irregular replacement of the cooling liquid and/or ageing of the cooling liquid lead to corrosion and premature failure of the water pump and other engine parts. The additives in the coolant agent function as ageing stabilisers, corrosion protection, anti-foam agent, detergents and coating material. All additives ensure the proper function and condition of the cooling liquid in accordance with the regulations until the next change.
Some of the most important functions and facts regarding coolant agent are stated below.
 |
ATTENTION |
Anti-freeze function of the coolant agent
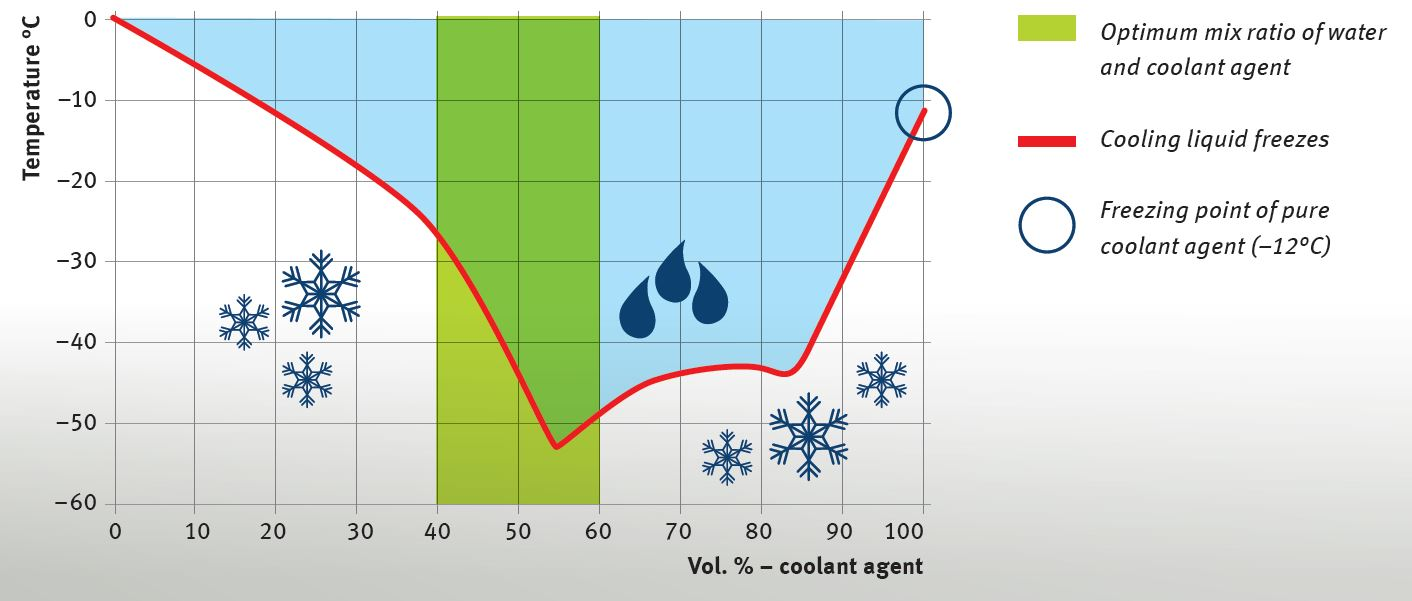
The main component of the coolant agent is monoethylene glycol, which has a very low freezing point. The cooling liquid used in the cooling system consists of a mixture of pure coolant agent and water that must be produced in a particular ratio in accordance with the engine manufacturer‘s instructions. A frequently used mix ratio is 50:50.
Undiluted coolant agent must not be used, even in areas in which very low frost temperatures are possible. If the coolant agent is mixed with an insufficient amount of water or if undiluted coolant agent is used, the anti-freeze effect reverses at a certain temperature. This means that the cooling liquid can freeze even at temperatures above –15°C, despite the high concentration of coolant agent.
The main component of the coolant agent is monoethylene glycol, which has a very low freezing point. The cooling liquid used in the cooling system consists of a mixture of pure coolant agent and water that must be produced in a particular ratio in accordance with the engine manufacturer‘s instructions. A frequently used mix ratio is 50:50.
Undiluted coolant agent must not be used, even in areas in which very low frost temperatures are possible. If the coolant agent is mixed with an insufficient amount of water or if undiluted coolant agent is used, the anti-freeze effect reverses at a certain temperature. This means that the cooling liquid can freeze even at temperatures above –15°C, despite the high concentration of coolant agent.
Thermal absorption capacity of the coolant agent
Pure coolant agent has a lower thermal absorption capacity than normal water. This means that a 50:50 mixture of coolant agent and water is unable to transport as much heat to the radiator as normal water. The engine manufacturer has taken this reduced thermal absorption capacity of the coolant agent into account in designing the cooling system. The circulating velocity of the water pump, the size of the radiator and the amount of cooling liquid have been adjusted accordingly. If coolant agent is mixed into the cooling liquid and the vehicle radiator has sufficient dimensions, then the engine itself is protected against overheating even in hot areas.*
Engines that are improperly operated using pure water may never reach the correct operating temperature as this means that the cooling system is oversized. For more detailed information on this topic, see Chapter ‚Damage and causes of failure‘.
Increasing the boiling point
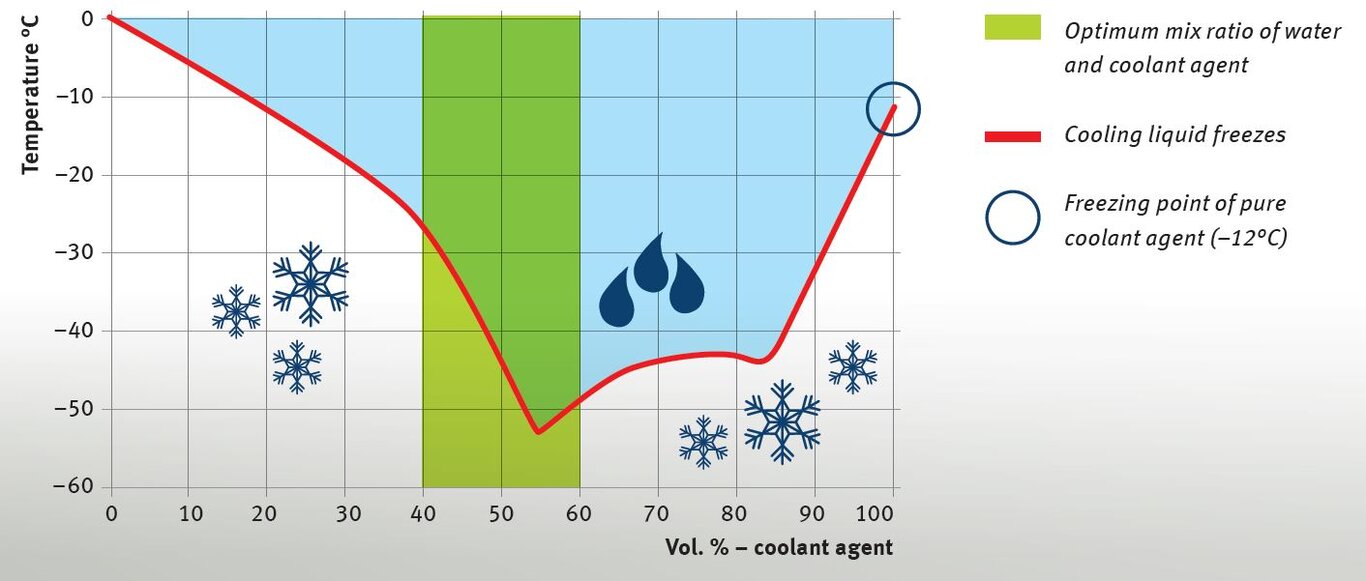
The boiling point of cooling liquid increases when the proportion of coolant agent is raised. At the level of air pressure present at sea level, pure water has a boiling point of 100 °C. For pure coolant agent based on monoethylene glycol, the boiling point is over 160 °C. The proportion of coolant agent therefore has a considerable influence on the boiling point of the cooling liquid. This means that the cooling liquid will only reach the boiling point at considerably higher temperatures in accordance with the proportion of coolant agent. This acts as a safety reserve in order to prevent cavitation on the engine components. The operating pressure in the cooling system (approx. 1 bar) further increases the boiling point.
The graphic shows the vapour pressure curves of certain glycol/water mixtures. The resulting boiling points can be read out at the respective intersections, for example at a operating pressure of 1 bar in the cooling system and with various mix ratios.
* In the case of used vehicles (facility vehicle) that were sold from temperate latitudes to hot climate zones, the size of the vehicle radiator may need to be adjusted in accordance with the manufacturer‘s instructions in order to prevent the engine from overheating. This is something that cannot be effectively prevented by operating the cooling system with pure water and/or with the thermostat removed.
Pure coolant agent has a lower thermal absorption capacity than normal water. This means that a 50:50 mixture of coolant agent and water is unable to transport as much heat to the radiator as normal water. The engine manufacturer has taken this reduced thermal absorption capacity of the coolant agent into account in designing the cooling system. The circulating velocity of the water pump, the size of the radiator and the amount of cooling liquid have been adjusted accordingly. If coolant agent is mixed into the cooling liquid and the vehicle radiator has sufficient dimensions, then the engine itself is protected against overheating even in hot areas.*
Engines that are improperly operated using pure water may never reach the correct operating temperature as this means that the cooling system is oversized. For more detailed information on this topic, see Chapter ‚Damage and causes of failure‘.
Increasing the boiling point
The boiling point of cooling liquid increases when the proportion of coolant agent is raised. At the level of air pressure present at sea level, pure water has a boiling point of 100 °C. For pure coolant agent based on monoethylene glycol, the boiling point is over 160 °C. The proportion of coolant agent therefore has a considerable influence on the boiling point of the cooling liquid. This means that the cooling liquid will only reach the boiling point at considerably higher temperatures in accordance with the proportion of coolant agent. This acts as a safety reserve in order to prevent cavitation on the engine components. The operating pressure in the cooling system (approx. 1 bar) further increases the boiling point.
The graphic shows the vapour pressure curves of certain glycol/water mixtures. The resulting boiling points can be read out at the respective intersections, for example at a operating pressure of 1 bar in the cooling system and with various mix ratios.
* In the case of used vehicles (facility vehicle) that were sold from temperate latitudes to hot climate zones, the size of the vehicle radiator may need to be adjusted in accordance with the manufacturer‘s instructions in order to prevent the engine from overheating. This is something that cannot be effectively prevented by operating the cooling system with pure water and/or with the thermostat removed.
Corrosion protection
The most important task of the coolant agent is to protect the cooling system against corrosion, which primarily affects the durability of the entire engine.
Due to a lack of corrosion-inhibiting substances in the cooling liquid, the salts and acids that may be present in the cooling liquid lead to components being chemically attacked (corrosion). In the long term, this deteriorates the engine components. Aluminium corrosion is a common problem in cooling systems in particular.
The oxygen present in the water also oxidises with ferrous materials and pollutes the cooling liquid with solids (rust). The relatively hard rust particles lead to rapid wear on the sliding ring seal of the water pump.
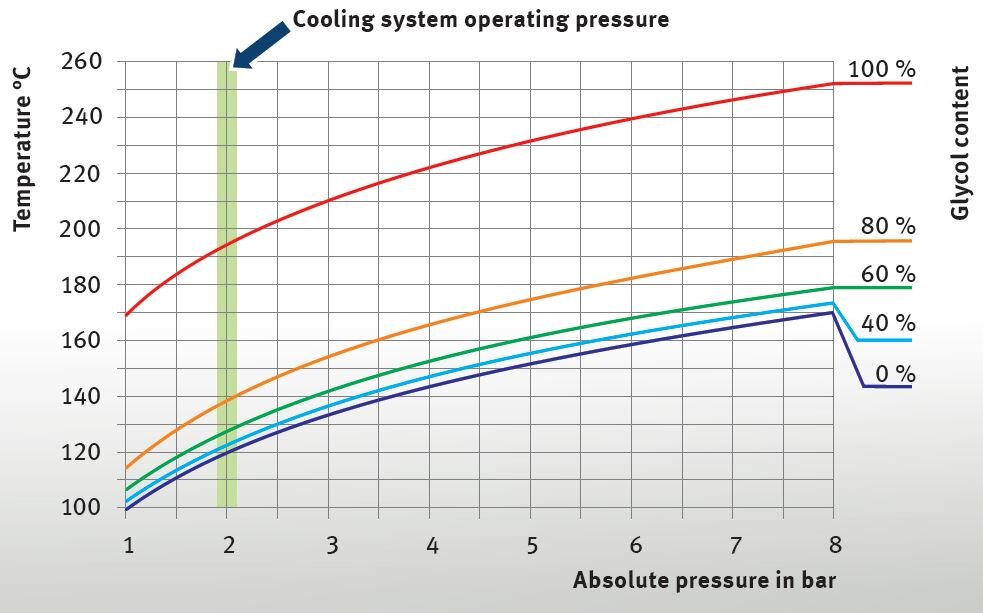
In order to counteract the corrosion, the coolant agent has alkaline properties. The pH value is around 8, providing a buffer effect with regard to acids that enter the cooling system. The buffer effect is decreasing
over time. Salty water, rainwater, deposits of radiator decalcifiers or combustion gases that enter the cooling liquid can move the ratio of acids to bases into the acidic range. Pure (distilled) water has a pH value of 7 and therefore features neutral properties.
The graphic shows the individual pH value range covered by the various example liquids.
The most important task of the coolant agent is to protect the cooling system against corrosion, which primarily affects the durability of the entire engine.
Due to a lack of corrosion-inhibiting substances in the cooling liquid, the salts and acids that may be present in the cooling liquid lead to components being chemically attacked (corrosion). In the long term, this deteriorates the engine components. Aluminium corrosion is a common problem in cooling systems in particular.
The oxygen present in the water also oxidises with ferrous materials and pollutes the cooling liquid with solids (rust). The relatively hard rust particles lead to rapid wear on the sliding ring seal of the water pump.
In order to counteract the corrosion, the coolant agent has alkaline properties. The pH value is around 8, providing a buffer effect with regard to acids that enter the cooling system. The buffer effect is decreasing
over time. Salty water, rainwater, deposits of radiator decalcifiers or combustion gases that enter the cooling liquid can move the ratio of acids to bases into the acidic range. Pure (distilled) water has a pH value of 7 and therefore features neutral properties.
The graphic shows the individual pH value range covered by the various example liquids.
Tags
Product groups
This might also interest you


